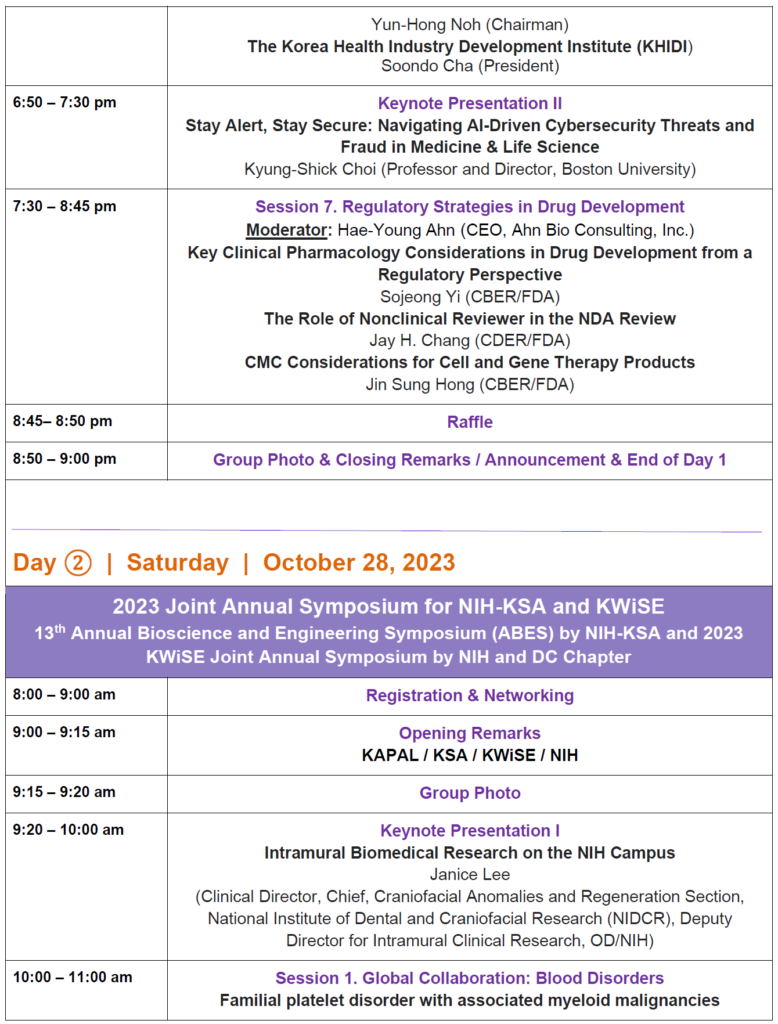
The 8th KAPAL Annual Conference, “Drug Development A to Z” with KIC-DC, KSEA-DC, and KLAM
안녕하세요.
다가오는 11월 1일(금요일)과 2일(토요일) 양일에 걸쳐 개최되는 한미생명과학인협회(KAPAL) 연례 컨퍼런스에 여러분을 초대합니다.
올해의 주제는 “Drug Development A to Z”로 신약 또는 의료기기를 개발하는 과정 전반에서 고려해야 할 부분들에 대해 전문가들의 의견을 듣고 논의하는 자리를 준비하였습니다. 또한, 미국 내 바이오산업 투자동향과 미국 내 제 3의 바이오허브인 메릴랜드와 버지니아 지역에서 제공하는 다양한 혜택에 대해 알아보는 시간을 가질 것입니다.
올해의 연례행사의 가장 큰 목표는 다양한 분야 및 단체와의 협력입니다. 한국혁신센터(KIC), 재미한인과학기술자협회 DC지부(KSEA-DC) 그리고 메릴랜드 한인 생명 과학자 모임(KLAM)과 함께 세부 프로그램을 기획하고 있어 더욱 다채로운 행사가 될 것입니다.
제 8회 KAPAL 연례컨퍼런스에서 다룰 주제는 다음과 같습니다.
- An overview of drug development
- Design of clinical trials and its operations
- An overall investment market in life sciences
- Legal issues for the foreign companies in the US
- Startup pitch contest- organized by Korea Innovation Center (KIC-DC)
- Startup incubation at Johns Hopkins- organized by Korean Life Scientist Association of Maryland (KLAM)
- Drone competition- organized by Korean-American Scientists and Engineers Association (KSEA-DC)
KAPAL 연례학회는 한미 양국의 기관 및 기업의 지원을 받아 매년 개최되고 있습니다. 특히, 주미대사관, 한국보건산업진흥원(KHIDI), 메릴랜드주의 지속적인 후원과 그라디언트 바이오컨버전스, SK바이오팜, 유한양행 USA, 휴온스 USA, 소마젠, 한국제약바이오협회, JW중외제약, 한올바이오, Reed Smith와 같은 기업 및 기관에서 후원을 결정해주셨습니다.
행사에 대한 상세 내용은 홈페이지(www.kapal.org)를 통해 지속적으로 업데이트할 예정입니다. 질문이나 건의사항이 있으신 경우, contact@kapal.org로 문의바랍니다.
행사 등록 링크: https://KAC2024.eventbrite.com
*KAPAL 회원이 되시면 컨퍼런스 등록시 등록비 할인을 받으실 수 있습니다. (할인코드: KAPAL)
(회원등록: KAPAL Registration)
**컨퍼런스 현장에서 재미한인과학기술자협회(KSEA, www.ksea.org) 회원가입을 무료로 지원할 예정입니다.
***참석하신 분들 중 추첨하여 소정의 기념품을 증정할 예정입니다.
Hotel information:
- Gaithersburg Marriott Washingtonian Center (예약링크: Reservation-Link (marriott.com))
- Residence Inn Gaithersburg Washingtonian Center (9721 Washingtonian Blvd, Gaithersburg, MD 20878)
- SpringHill Suites Gaithersburg (9715 Washingtonian Blvd, Gaithersburg, MD 20878)
——————————————————————————————————
Dear KAPAL members,
Greetings!
We would like to invite you to the 8th annual Korea-American Professional Association in Life Sciences (KAPAL) Conference, which will be held over two days, Friday, November 1st, and Saturday, November 2nd, 2024.
The theme for this year’s conference is “Drug Development A to Z,” where we will explore the various aspects that need to be considered throughout the process of developing new drugs or medical devices. Experts will be sharing their insights, and we will also discuss current investment trends in the U.S. biotech industry, along with the various benefits offered by Maryland and Virginia, the third-largest biotech hubs in the U.S.
The primary goal of this year’s event is fostering collaboration across various fields and organizations. We are co-organizing the program with the Korea Innovation Center (KIC), the DC Chapter of the Korean-American Scientists and Engineers Association (KSEA-DC), and the Korean Life Scientist Association of Maryland (KLAM), ensuring a more diverse and exciting event.
The key topics for the 8th Annual KAPAL Conference are as follows:
An overview of drug development
Design and operations of clinical trials
Investment market trends in life sciences
Legal issues for foreign companies in the U.S.
Startup pitch contest – organized by Korea Innovation Center (KIC-DC)
Startup incubation at Johns Hopkins – organized by Korean Life Scientist Association of Maryland (KLAM)
Drone competition – organized by Korean-American Scientists and Engineers Association (KSEA-DC)
The 8th KAPAL Annual Conference is made possible with the support of institutions and companies from both Korea and the U.S. Notably, the Korean Embassy in the U.S., the Korea Health Industry Development Institute (KHIDI), and the State of Maryland have been ongoing sponsors. This year, we also have sponsorship from companies and organizations such as Gradiant Bioconvergence, SK Biopharmaceuticals, Yuhan USA, Huons USA, Psomagen, Korea Pharmaceutical and Bio-Pharma Manufacturers Association, JW Pharmaceutical, HanAll Biopharma, and Reed Smith LLP.
We will continue to provide updates regarding the event on our website (www.kapal.org). For any questions or suggestions, feel free to contact us at contact@kapal.org.
Event registration: https://KAC2024.eventbrite.com
*KAPAL members will receive a discount on conference registration (discount code: KAPAL).
KAPAL Registration link: KAPAL Registration
**We will be offering free membership registration for the Korean-American Scientists and Engineers Association (KSEA,www.ksea.org) at the conference.
***There will be a raffle for attendees with a chance to win special souvenirs.
Hotel Information:
- Gaithersburg Marriott Washingtonian Center (Reservation-Link (marriott.com))
- Residence Inn Gaithersburg Washingtonian Center (9721 Washingtonian Blvd, Gaithersburg, MD 20878)
- SpringHill Suites Gaithersburg (9715 Washingtonian Blvd, Gaithersburg, MD 20878)
We look forward to your participation!