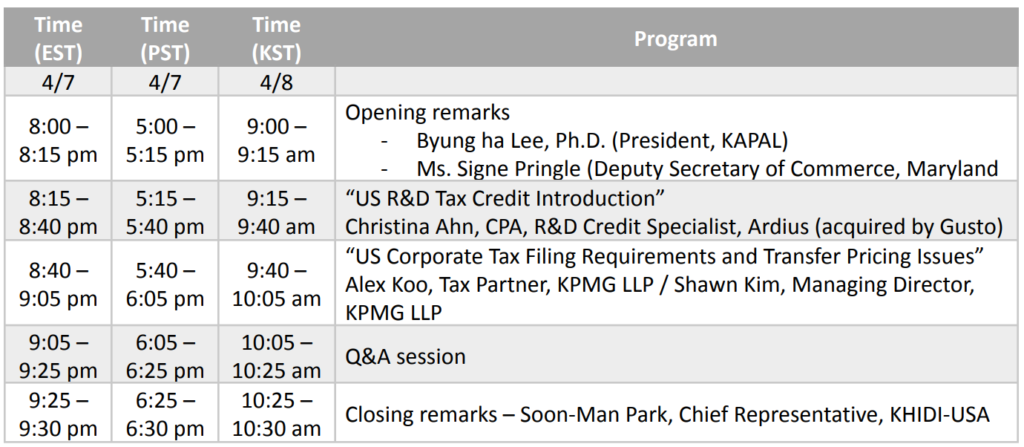
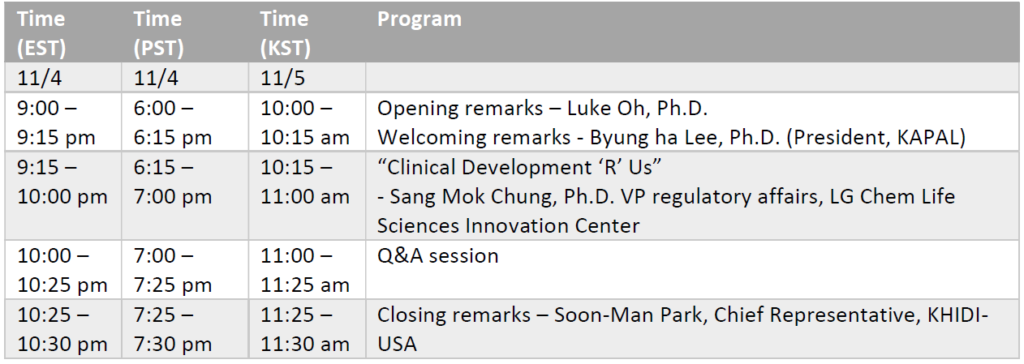
본 행사는 주 미국 대한민국 대사관에서 주최하며 한국보건산업진흥원 및 한미생명과학인협회 (캘리포니아)에서 주관하는 온라인 웨비나 및 오프라인으로 동시 진행되는 행사 입니다.

이벤트 신청 페이지: https://www.eventbrite.com/e/349092704677
연사 소개
발표 I : 박성원 변호사 (Sung Park) / Reed Smith LLP
Sung provides counsel to FDA-regulated companies in developing, distributing, and marketing FDA-regulated digital health products and, when necessary, in responding to regulatory and administrative enforcement actions by federal and state agencies. Sung frequently counsels clients on FDA’s legal and regulatory requirements and policies for digital health products, and provides counsel that allows companies to comply with FDA’s policies and requirements. Sung has experience in responding to FDA’s various enforcement actions such as warning letters, untitled letters, and form 483 notice of inspectional observations. Sung understands the unique business landscape of FDA-regulated products, and provides practical advice that suits the needs of the client’s business.
발표 II : 윤학희 CEO (Harry Yun) / InBody USA
Harry Yun is the CEO of InBody USA, a prominent medical device company from Korea and a worldwide leader in body composition technology with a mission to provide biomedical technology that simplifies the understanding of health and wellness. InBody USA was established in the year 2000 in Beverly Hills, California and has moved its corporate office to Cerritos, California in 2012. Under Yun’s leadership, InBody continues to successfully penetrate the US market with state-of-the-art technology. Yun is a graduate from Hankuk University of Foreign Studies (HUFS) with Masters in Economics and has extensive experience of International Marketing.
발표 III : 조성환 CTO (Sunghwan Cho) / NanoCellect Biomedical Inc
Sunghwan Cho is the CTO of NanoCellect Biomedical Inc, whose mission is to facilitate breakthrough biomedical discoveries by making cell analysis and sorting technology more affordable and easier to use. He has 15+ years of expertise in integrating microfluidics, optics, micro/nano-fabrication, and electronics for advanced biomedical devices and instruments. He is also an inventor of the company’s core technologies, including its microfluidics-based cell sorting platform, and manages the company’s research project and product development. He was selected as a Marylou Ingram Scholar of the International Society for Advancement of Cytometry (ISAC) from 2012 to 2017, recognized for his contribution to the field of cytometry.